Introduction to the Principle of Light Emitting Diode (LED: Light Emitting Diode)

Light-emitting diodes (LEDs) are all made of "compound semiconductors". The semiconductors formed by the bonding of two or more elements are called "compound semiconductors." For example: Gallium arsenide (GaAs) belongs to the third and fifth group compound semiconductors (3A group gallium and 5A group arsenic), cadmium selenide (CdSe) belongs to the 26 group compound semiconductor (2A group cadmium and 6A group selenium), etc. Solid materials and compound semiconductors have excellent luminous efficiency, so we mostly use them to make light-emitting components. For example, gallium arsenide (GaAs) belongs to the "direct band gap", so gallium arsenide wafers are made The components will emit light, and are generally used to make light-emitting diodes (LED), laser diodes (LD) and other light-emitting components.
The structure of the light-emitting diode (LED) is shown in Figure 1(a). The straight-inserted lamp bead has an oval appearance and the size is similar to that of a mung bean, but the real light-emitting part is only the "chip" in the picture. The size of the chip is similar to that of a grain of sand on the beach. A chip of this size can emit very strong light. Because the light-emitting diode chip is small, a 2-inch GaAs wafer can produce tens of thousands of chips. After dicing, it is packaged to form the appearance shown in Figure 1(a). The manufacturing process of light-emitting diodes is similar to that of silicon wafers. They are all made by photolithography, doping technology, etching technology, and thin film growth.
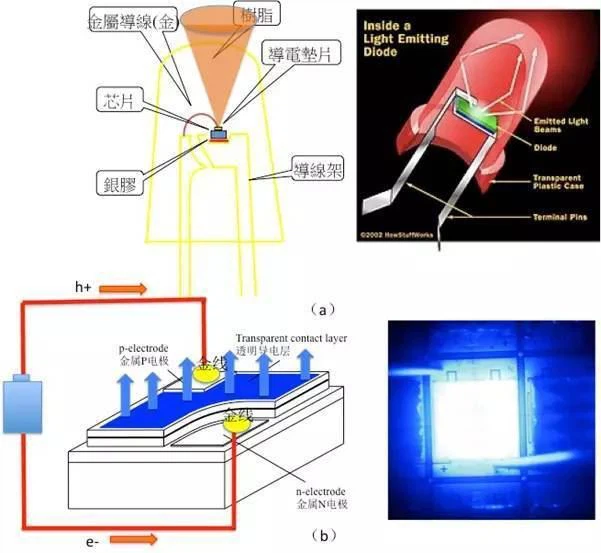
The basic principle of light-emitting diodes
If we enlarge the chip of the diode, as shown in the gallium nitride light-emitting diode in Figure 1(b), there are metal electrodes, and there are N-type and P-type gallium nitride and electrodes in the middle. When the light-emitting diode is connected to the battery, the electrons The negative electrode of the battery flows into the N-type semiconductor, and the holes flow into the P-type semiconductor from the positive electrode of the battery. The electrons and holes combine at the junction of the P-type and the N-type, and emit light from the top of the chip, passing through the oval plastic package shell. , Because the elliptical plastic packaging shell is similar to a convex lens, it has a light-gathering effect, which can make the emitted light "more concentrated".
It is worth noting that the only semiconductor components that can condense the emitted light into a beam are "laser diodes (LD)". To condense the light into a beam, a "cavity" structure is required. Regarding the laser, I will give a detailed introduction later.
LED color
When we apply voltage to different compound semiconductor materials, the compound semiconductor will emit "different colors of light." Scientists can use this principle to make light-emitting components of different colors. As shown in Table 1, the brief description is as follows:
Epitaxy method: refers to the method of growing compound semiconductors. "Liquid Phase Epitaxy" (LPE: Liquid Phase Epitaxy) is the use of heating to melt the compound semiconductor into a liquid, and then slowly cool it to form a solid single crystal structure; "Organic Chemical Vapor Deposition ( MOCVD: Metal Organic Chemical Vapor Deposition" uses organic metals and gases to spray directly onto GaAs wafers to form single crystal films (epitaxial).
Luminous color: refers to the color emitted by the light-emitting diode observed by the naked eye.
Silicon Carbide (SiC): The light-emitting color is "blue-green". Since there were no light-emitting diodes that can emit blue light in the early days, silicon carbide (SiC) is mostly used as blue light diodes, but the color emitted by silicon carbide is not real Blue, and the life of the device is not long (the brightness will gradually weaken), and the display that can play real images outdoors must be composed of the three primary colors of red, green, and blue. The early display does not have blue (because there is no blue light) Light-emitting diodes), so it can only play a monochrome screen (display text or simple graphics), but can not play real images.
Gallium Nitride (GaN):
It was not until 1995 that the team of Dr. Shuji Nakamura of Nichia Chemical Company developed a "gallium nitride (GaN)" light-emitting diode, which can emit blue light and has a long lifespan. However, the atoms of gallium nitride and gallium arsenide There is a lot of difference in size (lattice mismatch), so it cannot be grown on a "gallium arsenide substrate" and must be grown on a "sapphire substrate (alumina single crystal)".
Since the original price of sapphire crystal substrate was very high, the hardness was high and it was not easy to process, the cost was high, and many related patents were in the hands of Japan Nichia Chemical Company. The patent license caused the price of blue light diodes to be very high. Therefore, the price of blue LED in the early stage remained high. With Taiwan and the mainland investing in this technology, the current price of blue LED has been very low, and it is widely used in lighting and display products.
Group 26 "two-element" compound semiconductors: light-emitting diodes (single) including cadmium selenide (CdSe), zinc telluride (ZnTe), cadmium sulfide (CdS), zinc selenide (ZnSe), zinc selenide (ZnS), etc. Crystal solids) due to the short lifetime of the devices, so they are rarely used at present, but the "polycrystalline powders" of these materials are commonly known as "phosphors", and are currently widely used in traditional electronic picture tube displays, plasma displays, and white light emitting diodes. And other products. In addition to group 26 compound semiconductors, scientists have also successively developed many phosphors with different compositions to increase the brightness and service life of the light.
Center wavelength of light-emitting diode
"Central wavelength" refers to the relative emission wavelength of the color emitted by the light-emitting diode. Since different colors of light have different wavelengths, different colors emitted by the light-emitting diode will have a relative emission wavelength. Take the "aluminum phosphide (AlP)" light-emitting diode as an example. The color seen by the naked eye is "green", but its emission spectrum is shown in Figure 2. It can be seen from the figure that the aluminum phosphide (AlP) light-emitting diode emits The color of light ranges from 0.45μm (blue-green) to 0.55μm (yellow-green), so it is not true green, but a color that is a mixture of many wavelengths, but the center wavelength of 5.0μm (green) has the highest light intensity. Therefore, the color seen by the naked eye is "green", and the wavelength with the strongest luminous intensity is called the "central wavelength". In other words, as long as you know the center wavelength of the light emitted by the light-emitting diode, you know what color it looks like to the naked eye.
The spectrum emitted by light-emitting diodes has a wavelength range (0.45μm~0.55μm), which is called "light impure". We can also use the Full Width Half Maximum to measure the purity of light. The component that emits "very pure light" is called "Laser".
Types of light-emitting diodes
If it is classified by chip structure, it can be divided into formal chip, flip chip and vertical structure chip. As shown in Figure 3(a), the structure where the positive and negative (PN) electrodes are on the same light-emitting surface is called a positive mounting structure. Since no suitable conductive substrate for gallium nitride material can be found, it has been successfully developed and industrialized in blue light. Since then, sapphire has always been the most important substrate material for this structure. Since the substrate material is not conductive, the general blue, green and violet gallium nitride LEDs are all of this structure.
The earliest red-yellow LED structure uses conductive gallium arsenide and gallium phosphide substrates, so they are all made into a single-electrode vertical structure. This structure is the earliest LED structure. Due to the good uniformity of light and current, The directivity is strong, so blue LEDs also began to use vertical structure LEDs in lamps for special applications. However, because the vertical structure LED process of gallium nitride material is more complicated and difficult, the sapphire substrate needs to be peeled off or the sapphire substrate needs to be peeled off directly. Gallium nitride grows directly on conductive silicon or metal substrates, so the yield is more difficult to control. Therefore, blue, green or purple LEDs with this structure are used in special applications, such as mobile phone flashes, flashlights with strong directivity, and car lights With UV curing lamps, in addition to the application of light needs, the high cost can only be used in high-end is also an important reason.
The flip-chip structure is shown in Figure 3(c). It is similar to the front-mounted structure. It is made of sapphire substrate material, but the light-emitting surface needs to be plated with reflective electrodes, and then flip-chip bonded to other substrates. The flip-chip emits light from the back , The positive and negative PN electrode surfaces reflect light. Because the heat transfer path is short and the heat transfer material is superior to sapphire, the flip-chip structure can have superior thermal stability and can drive higher current than the positive chip without attenuation.



